Is Your Organization Prepared for Risk-Based Inspections? Depends on Who You Ask.
With the adoption of ICH E6 (R2), risk-based quality management for clinical trials has become an expectation, and inspectors are starting to use these guidelines in their audits. However, The Avoca Group’s research, documented in the 2017 Avoca Industry Report, indicates that sponsors and providers both have work to do to be truly prepared for inspections in light of ICH E6 (R2).
In Avoca’s survey, sponsors and providers were asked how familiar they were with risk-based approaches to inspection preparedness. But – this is key – responses were categorized based on whether the respondent was a member of the Quality department or was in Clinical Operations. Across sponsors and providers, Quality personnel indicated a significantly higher level of knowledge about risk-based inspection preparedness relative to those in Clinical Operations.
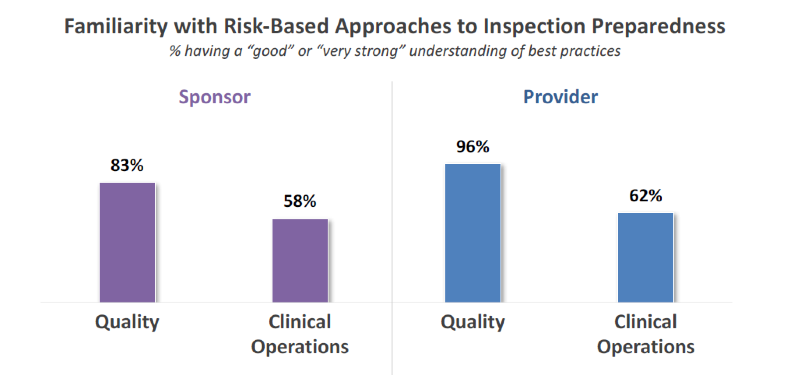
In other words, Quality is feeling really good about the new approaches to inspection preparation, but Clinical Operations is less confident and fluent. There are several contributing factors to this remarkable gap in self-assessed familiarity.
First, there is departmental friction: an ugly truth, but one that must be faced. Historically, Quality and Clinical Operations have not always had the best working relationship. Clinical Operations has often perceived Quality as a “policing” organization – the equivalent of someone coming in after you have cleaned the house from top to bottom and saying, “You missed a spot.” The result? Clinical Operations may not be particularly inclined to proactively collaborate with Quality, so may not be receiving the full benefit of Quality’s knowledge about risk-based inspection procedures.
Second, the two departments may have contrasting perspectives on who is responsible for the success of an inspection. Clinical Operations may view managing inspections as “Quality’s job.” However, while Quality is responsible for preparing for and hosting the inspection, the work under review has all been done by Clinical Operations. Both departments, therefore, are jointly responsible for a successful inspection.
Third, there may be an unconscious bias on the part of Clinical Operations that they don’t need to know best practices about risk-based approaches to inspection preparedness because Quality will tell them what they need to do when an inspection is foreseen. This sets the organization up for increased risk, because Clinical Operations may not proactively maintain a constant state of inspection readiness throughout the clinical trial’s process.
The gap in inspection preparedness can be closed – bringing both Quality and Clinical Operations up into the 90% range and higher – once both departments acknowledge that maintaining a constant state of inspection preparedness is everyone’s job. But to do this job, these departments need to partner together much more closely than ever before. Quality needs to systematically and conscientiously share its knowledge of best practices and inspection procedures with Clinical Operations. Clinical Operations should confer with Quality on a regular basis to optimize processes in line with ICH E6 (R2).
Success will be readily evident. When Quality and Clinical Operations partner together effectively, Clinical Operations as well as Quality will express confidence toward inspection preparedness – and the inspection itself will be passed with flying colors.
Is your organization inspection ready? The Avoca Group specializes in supporting sponsors and CROs to comply with ICH E6 (R2) through the use of risk-based approaches to quality and oversight. The Avoca Group’s consulting services are grounded in the leading practices and tools from the Avoca Quality Consortium® and customized to drive measurable improvements to quality and execution of clinical trials. To learn more, or to speak with a representative, contact us.
Download The 2017 Avoca Industry Report: Using Risk-Based Approaches to Inspection Preparedness
.
Author:
 |
Dennis Salotti
Vice President, Operations, The Avoca Group |
The Avoca Group’s Consulting Services for Inspection Readiness provide full management through point-of-need support for inspections. Learn more >> |

