Bridging the Gaps in CAPA Planning in Clinical Trials
May 2020
When it comes to implementing corrective and preventive action (CAPA) plans in clinical tri-als, we consistently hear similar frustrations from members of the research community year after year. They typically identify and implement corrective actions, but identifying and implementing preventive actions, which aim to minimize risks to other studies, is still somehow getting lost in the shuffle.
Even with all the regulations, related guidelines, trainings, company policies, procedures, and quality systems in place, the same issues keep reoccurring in different trials. This happens for several reasons, namely because, when it comes to root cause analyses (RCAs), the correct root causes often aren’t identified beyond the individual study being reviewed. More often than not, the focus is solely on the tactical issue or issues at the study level, rather than also at the system level, or across studies. Consequently, issues are frequently only resolved within the individual study where they were identified, in a tactical way, and not further addressed with preventive measures for other, ongoing, and future clinical trials. A key reason RCAs are frequently not incorporated at the system level is the lack of available data on issues across trials. This makes pinpointing the systemic root cause(s) very difficult and frustrating for organizations that see the same issues and challenges —believed to have been previously addressed—pop up across their portfolio in other trials.
Getting to the root of the problem
In 2018, the International Council for Harmonization (ICH) of Technical Requirements for Pharmaceuticals for Human Use issued an addendum to update its guideline for good clinical practice (GCP). Taking the evolution of new technology and use of e-clinical tools into consideration, ICH E6 (R2), as the addendum is commonly known, encourages life sciences organizations to pursue innovative approaches for conducting clinical trials, specifically quality by design (QbD) and risk-based quality management (RBQM). ICH E6 (R2) also set the expectation that sponsors and contract research organizations (CROs) have systems in place to prevent non-compliance and efficiently manage significant deviations with appropriate CAPAs. However, regulatory findings worldwide consistently reveal that the CAPAs that sponsors, CROs, and investigators have in place are insufficient because there isn’t enough focus on preventive measures at the system level, across their studies.
In clinical research, quality issues that come up must be addressed and resolved in a timely, effective, and regulatory compliant manner. When a problem is minor and can be immediately addressed with a sufficient solution, then the quality issue can be easily resolved. However, sometimes issues are more significant and complex, and require a CAPA plan. When developing a CAPA plan, the quality issues that need to be addressed may be isolated or more broadly represented across a clinical trial or entire R&D programs. Either way, these issues can create risks to clinical trial data integrity and/or patient safety.
To assist with systemic RCA, we should ensure data and metrics on issues across trials are readily available.
The missing link
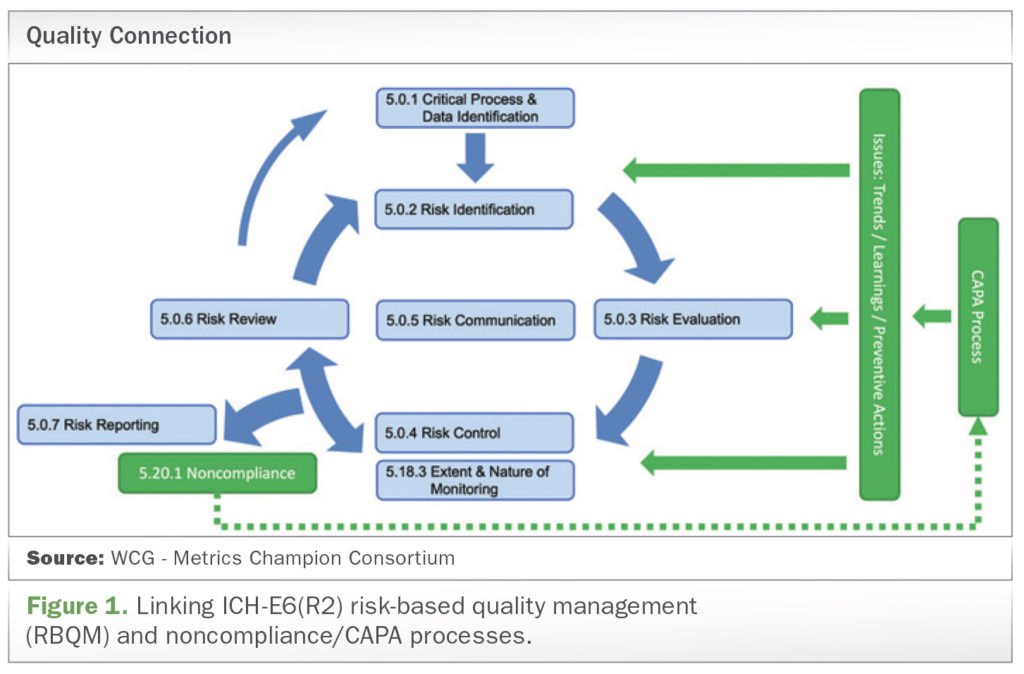 Among the most significant changes related to ICH E6 (R2) is the introduction of guidance on a new risk-based approach to the management of quality in clinical research. The updated guideline calls for a formal approach to quality management that embraces technology and leverages access to real-time data to drive a more structured approach to risk management. Specifically, in ICH E6 (R2) Section 5.0, quality management now includes the efficient design of a trial to avoid unnecessary complexity, procedures, and data collection.
Among the most significant changes related to ICH E6 (R2) is the introduction of guidance on a new risk-based approach to the management of quality in clinical research. The updated guideline calls for a formal approach to quality management that embraces technology and leverages access to real-time data to drive a more structured approach to risk management. Specifically, in ICH E6 (R2) Section 5.0, quality management now includes the efficient design of a trial to avoid unnecessary complexity, procedures, and data collection.
In ICH E6 (R2), the addendum to Section 5.20.1 specifies that “a sponsor should perform root cause analysis and implement appropriate corrective and preventive actions,” i.e., CAPA. However, a limiting factor is that the preventive actions mentioned here are not explicitly linked to the control and reduction of risk in Section 5.0 Quality Management.
As an established pathway to identify and implement certain preventive actions, RBQM is the missing ingredient in effective CAPA plan execution. This risk-based approach to the management of quality in clinical research helps to ensure that corrective and preventive actions tackle the root cause of problems and minimize their reoccurrence. That’s because the basic premise of RBQM is the identification of risks on a continuous basis throughout the lifecycle of a clinical trial, from design and conduct to evaluation and reporting. Application of RBQM approaches can facilitate better, more informed decision-making and more efficient utilization of available resources.
Imagine having access to all this information and knowing where things went wrong in the past and the conditions that led to the issues. The value of that knowledge cannot be taken for granted and RBQM makes it tangible. For example, when RBQM is part of the process from the initial stages, risk mitigation can inherently be built into the protocol. Furthermore, in this way, issues related to standard operating procedures (SOPs) and underlying conditions can be mitigated or proactively monitored, minimizing opportunities that lead to reoccurring problems.
Connecting the dots: Bringing CAPA and RBQM together
Similar to breaking down data silos, we need to break down the silos that exist between CAPA and RBQM teams. Within R&D organizations, CAPA management often sits on the quality assurance (QA) side of the business, while RBQM has evolved into its own multi-disciplinary area. In practice, it’s as though these groups reside in different countries, where they don’t speak the same languages. Perhaps what’s needed is as simple as a translator to help them communicate and, ultimately, work together. The FDA, the European Medicines Agency (EMA), and the ICH all advocate using a risk-based approach to monitoring data quality in clinical trials. While long touted as a more proactive way to reduce risk and improve data quality while streamlining costs, RBQM continues to be implemented in different ways, mainly because of the varying interpretations of the ICH E6 (R2) guidance. By using current and emerging issues to help us understand systemic root causes, we can implement preventive actions to reduce risk on current and future studies. After all, learning from current and emerging issues is a critical part of RBQM.
While the changes in ICH E6 (R2) affect many clinical research stakeholders, the greatest impact continues to be on trial sponsors, who must ensure that the revisions are reflected in the processes and daily practice of study conduct. In bringing the RBQM language into ICH E6 (R2), there is an opportunity to improve CAPA processes through a clear link between applying learnings from current and emerging issues to risk control and reduction at the system level.
Connecting CAPA and RBQM in this way will enable real-time learning about existing risks, as they evolve, in addition to ongoing identification of emerging issues and potential threats. Armed with this continuous knowledge, sponsors and their CRO and vendor partners will be better equipped to reduce the reoccurrence of issues that repeatedly trigger CAPA processes, enabling more focused and efficient trial operations.
Linda B. Sullivan is Co-Founder MCC, WCG Avoca Senior Advisor
