The 2020 Avoca Quality and Innovation Summit took place virtually on October 14-15, 2020.
About the Event
Clinical trials have changed, will continue to evolve, and there is no going back in time. At our “Own the Future” virtual Summit, we will explore two distinct but complementary themes: the importance of quality as an underpinning of everything we do and innovation from the perspective of not only what we pursue with clinical trial execution but also from the standpoint of how we execute clinical trials.
Now in its ninth year, the Avoca Summit focuses on quality and the adoption of innovative technology in the execution of clinical trials. With two days of interactive sessions, panel discussions, and workshops, the Summit offers attendees the opportunity to engage with peers and thought leaders and to influence global change.
The collaborative nature of the event and diversity of participants allows for sharing of experiences and stimulating conversations. Individuals who work outside the clinical research space are able to view the important work of clinical trials through a distinctly different lens.
How will the Summit help you “Own the Future”?
- By collaborating in a deep-dive workshop on ICH E6 (R3), you will be part of the early work focused on what will be needed for organizations to successfully implement the upcoming regulatory changes.
- You will gain an understanding of how risk management strategies differ based upon the size of the organization, helping you to implement meaningful change within your organization.
- You will walk away with specific ideas for developing patient-centered drug development strategies, including hearing from patient themselves, that you can leverage within your organization.
- You will be inspired by thought leaders sharing their views on quality and innovation and the need for accelerating adoption of new technologies and operating models in clinical trial execution.
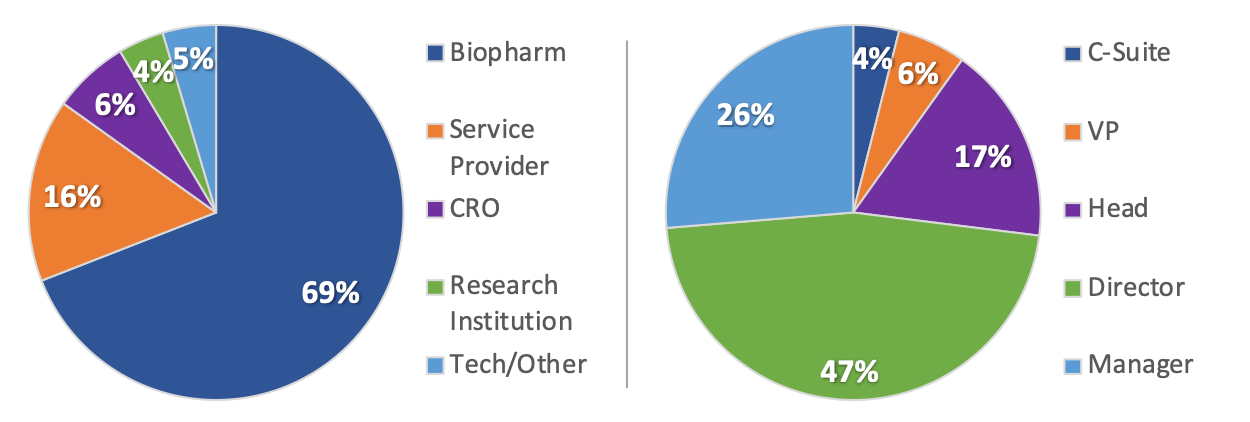
2019 Summit Attendee Profile
Last year’s attendees represented leadership positions from manager to C-Level from biopharma, clinical research, and tech companies across 10 countries.